Titanium tubing is increasingly integral to refinery reliability and efficiency, where corrosive media, variable temperatures, and strict uptime demands converge. Widely used in chemical processes such as chlor-alkali, salt production, urea, pesticides, synthetic fibers, organic synthesis, pharmaceuticals, and hydrometallurgy, titanium’s self-healing passive film provides outstanding resistance to chloride-induced pitting, crevice attack, and stress corrosion cracking. Its high specific strength supports thin-wall designs that reduce weight and pressure drop while maintaining pressure integrity.
In critical heat-transfer services, Titanium heat exchangers used in refineries employ titanium tubes to preserve stable heat flux in brines, sour water, and organic acids that rapidly degrade conventional alloys. Titanium is used to make heat exchange or condensation cooling equipment with serious corrosion in some key parts—such as overhead condensers, seawater-cooled units, and caustic coolers—extending service life and reducing unplanned downtime. With proven weldability and compliance to ASTM/ASME standards, titanium tubes help lower chemical dosing, simplify maintenance, and improve lifecycle economics across modern refining operations.
1. Corrosive environments for titanium materials in refining and petrochemical equipment
In refinery and petrochemical plants, titanium’s deployment must balance corrosion resistance with mechanical limits. From a mechanical performance standpoint, industrially pure titanium equipment should generally operate at temperatures not exceeding about 230°C, while titanium alloy equipment should not exceed about 300°C. Within these bounds, titanium excels in low-temperature (<230°C) light-oil corrosion environments commonly encountered in overhead and condenser services:
· H2S–HCl–H2O: Atmospheric/vacuum distillation overhead condensation and cooling systems exposed to wet HCl and H2S.
· H2S–HCN–H2O: Catalytic cracking absorber/stripper systems where cyanides and sulfides coexist with water.
· H2S–CO2–H2O: Claus tail gas or desulfurization regenerator overhead condensation systems with acid gases.
· H2S–CO2–RNH2–H2O: Amine solvent regeneration reboiler bottoms and associated coolers/condensers.
· H2S–NH3–H2O: Sour-water stripper condensers and coolers with ammonium salts and dissolved sulfides.
By pairing appropriate titanium grades with conservative temperature limits and sound fabrication, titanium tubing in Heat exchangers and coolers resists the aggressive chemistry that undermines copper alloys and austenitic stainless steels in these mixed-phase, salt-forming services.
1.1 Effect of cooling water on titanium tubes
Refinery operations rely heavily on cooling water networks—often using seawater, brackish water, or reclaimed wastewater. Titanium tubes demonstrate outstanding resistance to these corrosive cooling waters, maintaining passivity in chloride-rich streams and under biofouling pressures that accelerate pitting and crevice attack in conventional alloys. Whether configured as BWG20 Titanium tube or BWG22 Titanium tube, thin-wall titanium sustains clean heat-transfer surfaces, tolerates higher velocities without erosion-corrosion, and enables compact designs with lower pumping penalties.
1.2 Effect of hydrogen sulfide (H2S) on titanium tubes
Sulfur is present in crude oils predominantly as organosulfur compounds, with traces of dissolved H2S. Though upstream removal is limited, refining steps—heating, atmospheric/vacuum distillation, hydrotreating, and catalytic processes—transform organosulfur species into H2S. Titanium is notably resistant to sulfide attack and pitting in wet H2S at typical refinery coolant-side temperatures and is immune to sulfide stress corrosion cracking (SSCC). In many units processing high-sulfur crudes, overhead systems carry elevated H2S; here, titanium tubing in overhead condensers has shown superior durability. One refinery handling 3%–5% sulfur crude eliminated persistent overhead corrosion by adopting titanium for the top condensate cooling service.
1.3 Effect of sulfur dioxide (SO2)
SO2 arises in processes such as olefin sulfation/desulfation and in certain gas-treatment circuits. Titanium resists sulfide and SO2-related attack, including conditions where SO2 combines with condensate to form sulfurous/sulfuric acids that aggressively corrode carbon steel and many stainless steels. The self-healing oxide film on titanium hinders acid ingress and prevents under-deposit attack in condensing environments.
1.4 Effect of carbon dioxide (CO2)
CO2 exists in crude and natural gas or dissolves into wash and stripping waters. Wet CO2 corrosion, notorious in surface condensers used with amine-treated feedwaters, is a chronic problem for traditional materials. Titanium has excellent resistance to both dry and wet CO2. In gas-side separation zones of surface condensers, where steam condensate can become acidic, titanium maintains integrity, limiting metal loss and leakage that would otherwise compromise vacuum and product recovery.
1.5 Effect of ammonia (NH3)
Ammonia originates from the decomposition of organic nitrogen compounds in crude or is deliberately injected to neutralize acids. Titanium resists ammonia up to roughly 149°C; with adequate water present, passivity is sustained. Titanium performs reliably in crude unit overhead condensers and sour-water stripper ammonia overhead condensers. It is also essentially immune in boiling concentrated ammonium hydroxide solutions up to about 70% concentration, providing a margin of safety for upsets.
1.6 Effect of oxygen (O2)
Although oxygen is seldom a controlled constituent in most refinery streams, it often enters with feedstocks, by air ingress under vacuum, or via contact with aerated steam and wash water. Oxygen generally enhances titanium passivation, stabilizing the protective oxide and improving resistance in mixed-oxidant condensate environments. This attribute supports long-term stability in Heat exchangers and coolers undergoing frequent start-stop cycles and variable aeration.
1.7 Effect of hydrogen (H2)
Titanium is generally suitable up to approximately 315°C in environments with moderate hydrogen partial pressure where water or other passivators are present; however, in certain conditions, hydrogen embrittlement may occur. Risk factors include mechanical damage to the oxide film and excessive hydrogen uptake beyond approximately (800–900)×10^-4 wt%. Field experience and laboratory data indicate that maintaining a robust surface oxide reduces hydrogen ingress. Best practices include:
· Removing surface contamination—especially iron—from titanium before service by pickling in a room-temperature solution of 35 vol% HNO3 + 5 vol% HF for 3–5 minutes.
· Applying anodizing or controlled thermal oxidation to fortify the oxide film prior to hydrogen service.
Despite these mitigations, titanium is not recommended for pure hydrogen environments. Where hydrogen is present alongside water/steam and oxidants, titanium’s resistance remains strong, especially on the coolant side of refinery exchangers.
2. Applications of titanium in refining units
Historically, titanium first entered refineries as tubing for coolers, particularly in seawater-cooled services. As refining technology and cost optimization advanced, titanium found broader use beyond seawater exposure, proving effective even in process heat exchangers not cooled by seawater.
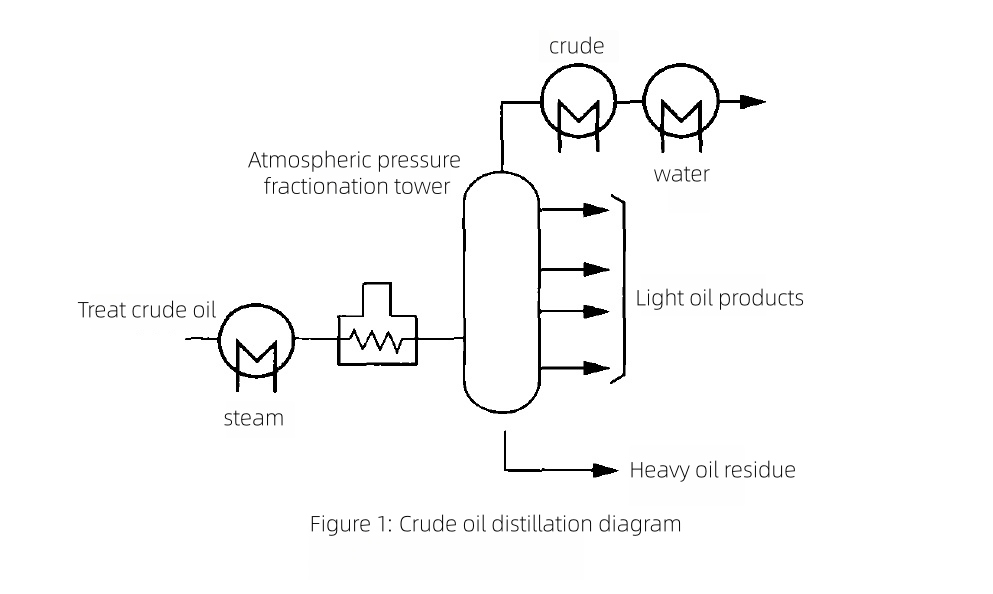
2.1 Crude distillation
In atmospheric and vacuum crude distillation, titanium tubing is used in overhead condensation and cooling systems to mitigate corrosion in HCl–H2S–H2O environments. High-boiling gas oils and residues are heated and fractionated to yield light fractions; the overheads contain chlorides and sulfides that condense with water, forming acidic brines. Titanium overhead condensers and air coolers prevent pitting, under-deposit corrosion, and chloride-induced stress-corrosion cracking that afflict copper alloys and austenitic stainless steels. Selecting BWG20 Titanium tube or BWG22 Titanium tube enables thin-wall, high-efficiency bundles with excellent service life.
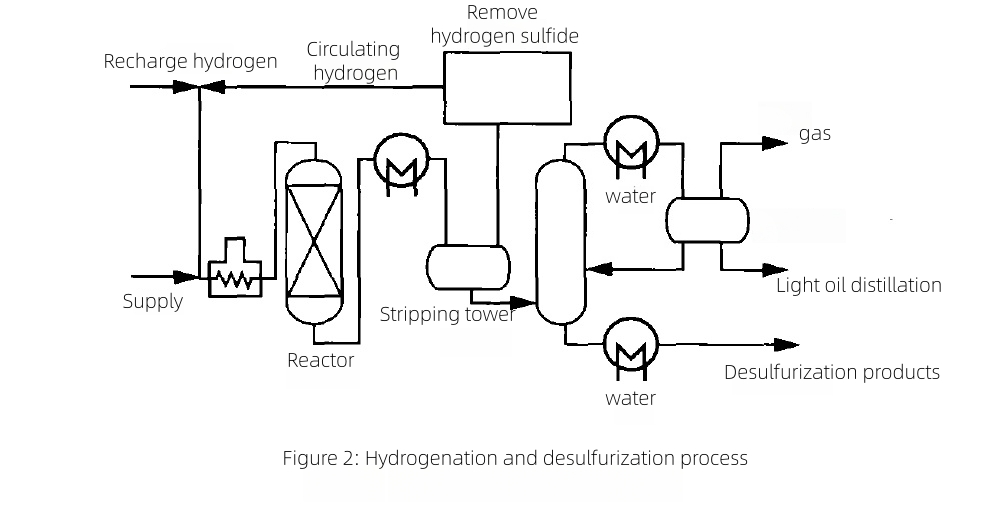
2.2 Hydrodesulfurization (HDS)
In HDS, feedstocks contact hydrogen over catalysts at temperatures reaching ~343°C, cracking organosulfur compounds to form H2S. Reactor effluent is cooled; residual hydrogen is separated from product vapors that can contain H2S, HCl, NH3, and steam before downstream fractionation. Titanium tubing is applied in reactor overhead condensers to thwart chloride and sulfide corrosion and in product/stripper coolers where wet acid gases condense. Its immunity to SSCC and resilience in wet H2S reduce leaks and extend run lengths.
2.3 Acid gas removal
Acidic gases (H2S, CO2) are commonly removed from refinery streams using aqueous amine solutions (e.g., MEA, DEA, MDEA). Titanium tubes have performed successfully in MEA reboilers, lean/rich exchangers, and overhead condensers, resisting wet CO2, amine degradation products, and oxygenated condensate. Titanium’s resistance to carbamate-induced corrosion and its stable passive film help maintain exchanger cleanliness and thermal performance.
2.4 Solvent extraction
Many refinery and petrochemical separations rely on solvent extraction with solvent recycle. Recycle concentrates trace acids, sulfides, chlorides, and degradation products, elevating corrosion risks in conventional metals. Titanium tube heat exchangers withstand these accumulations, suppressing pitting and crevice corrosion and preserving heat-transfer coefficients over long campaigns.
2.5 Fluid catalytic cracking (FCC)
In FCC ancillary equipment, titanium tube bundles have replaced traditional metals in fractionator condensers, secondary condensers, and debutanizer overhead condensers. Operating histories report excellent outcomes without detectable corrosion. Titanium’s performance advantage is particularly evident where wet H2S, NH3, HCN, and chlorides condense together, producing aggressive ammonium salt deposits that rapidly attack copper alloys and stainless steels.
3. Summary: Proven replacement for traditional alloys in corrosive, low-temperature refinery service
Across more than four decades, titanium has proven to be an exceptional substitute for Cu alloys, Cu–Ni alloys, and austenitic stainless steels in refinery Heat exchangers and coolers operating in low-temperature (<230°C) corrosive services containing H2S, Cl–, CN–, CO2, NH3, RNH2, as well as seawater and contaminated cooling waters. The combination of:
· Stable, self-healing passivation in mixed acid-gas condensates;
· Immunity to SSCC and strong resistance to wet H2S pitting;
· Excellent performance in wet CO2, ammonia-bearing waters, and solvent-recycle conditions;
· Thin-wall efficiency using BWG20 Titanium tube and BWG22 Titanium tube;
delivers longer run lengths, fewer leaks, lower chemical dosing, and improved lifecycle economics compared to traditional materials.
Frequently Asked Questions and Answers
Q1: What specific roles does titanium play in corrosion-resistant equipment within the refining industry, and how does it address challenges posed by harsh chemical environments (e.g., acidic catalysts, sulfur compounds)?
A1: Titanium serves as tubing and component material in Heat exchangers and coolers, overhead condensers, stripper condensers, and amine service exchangers. Its passive TiO2 film resists chloride, sulfide, cyanide, and acidic condensates, preventing pitting, crevice corrosion, and sulfide stress corrosion cracking. Titanium’s robustness in wet H2S, wet CO2, and ammoniated waters directly addresses the mixed-acid and salt-deposit environments that defeat copper alloys and many stainless steels.
Q2: How does titanium enhance the durability and operational efficiency of high-temperature/high-pressure components (e.g., reactors, heat exchangers) in petroleum refining processes compared to traditional materials like stainless steel or nickel alloys?
A2: Within recommended temperature limits (CP Ti ≤ ~230°C; Ti alloys ≤ ~300°C for equipment, and coolant-side services well below these), titanium sustains passivity under aggressive condensates, enabling thin-wall BWG20/BWG22 tubing with high heat-transfer efficiency and low pressure drop. It resists SSCC and under-deposit attack, reducing leaks and fouling. Fewer outages and stable U-values translate to higher on-stream factors and lower lifecycle costs than many stainless or Cu-Ni systems.
Q3: What role does titanium play in improving product purity and reducing maintenance downtime in the refining industry, particularly in processes involving corrosive feedstocks or catalytic reactions?
A3: Titanium minimizes metal ion contamination (e.g., Fe, Ni, Cu) into product and solvent circuits by resisting corrosion, preserving product purity and catalyst activity. Its resistance to salt-deposit corrosion in overhead systems reduces tube failures and cleaning frequency, extending turnaround intervals. In amine, sour water, and solvent-recycle services, titanium’s durability maintains condenser integrity and vacuum efficiency, cutting unplanned downtime and ensuring consistent product recovery.


