Titanium MRI have a long, In the field of medical imaging. commercially pure (CP) titanium—biocompatible, non‑ferromagnetic, and corrosion‑resistant—has become a go‑to for plates, screws, and joint hardware where imaging follow‑up is routine. Patients often ask, “Is titanium safe in MRI?” For non‑ferromagnetic implants like CP Ti, the answer is generally yes under MR‑Conditional labeling: minimal magnetic attraction, low RF heating, and reduced artifact compared with stainless steel. Typical density ~4.51 g/cm³ and elastic modulus ~105 GPa deliver strength with a closer bone stiffness match, helping comfort and longevity. In practice, Titanium MRI implants can be scanned at 1.5T and 3T with standard precautions, producing smaller susceptibility artifacts that preserve diagnostic detail around soft tissues and marrow.
Clinicians value Ti for its high fatigue resistance in physiological environments and its stable TiO2 passive film, which limits ion release. For radiologists, mri titanium hardware yields artifact reductions of 20–60% versus ferromagnetic alloys, improving postoperative assessment of tendon, nerve, and infection. Clear documentation remains essential: device model, MR‑Conditional limits (SAR, scan time, field strength), and body position. When properly selected and labeled, CP titanium supports safe imaging, accurate diagnosis, and reliable orthopedic outcomes. The Performance Multiplier in High-Performance Vehicles
How Metals Behave in Strong Magnetic Fields
· Mechanical forces: Ferromagnetic objects can experience torque and translational pull, risking shift or displacement.
· Induction and heating: Radiofrequency (RF) fields can induce eddy currents in conductive objects, causing temperature rise (the “thermal effect”), which depends on geometry, orientation, and pulse sequence.
1. Aneurysm Clips, Cranial Fixation Screws, Microcoils, and Vascular Stents
· Materials and magnetism: Modern devices are commonly made from titanium or titanium alloys; these are non-magnetic or weakly magnetic and, in most cases, are MR-Conditional or MR-Safe when used within labeled limits.
· Preoperative clarity: Before surgery, patients should confirm with their surgeon exactly which implant material and model will be used. After surgery, carry implant cards or documentation.
· Artifact awareness: While Grade 2 CP titanium largely eliminates concerns about displacement and heating, MRI “susceptibility” artifacts can persist—especially at higher field strength (3.0 T). If the follow-up region lies close to a metallic clip or coil, consider lower field imaging (≤1.5 T) and artifact-reduction protocols (SE over GRE, high bandwidth, thinner slices, optimized orientation).
Practical tips:
· Use 1.5 T when feasible for postoperative brain or neck imaging near metallic clips.
· Apply metal-artifact reduction sequences (MARS/SEMAC/MAVRIC) where available.
2. Dental Work and Dentures
· Removable dentures: If a prosthesis is removable, take it out before scanning to avoid artifact and ensure comfort.
· Fixed prosthetics: For fixed dental restorations, decision depends on proximity to the area of interest and artifact severity. Many titanium posts and implants are acceptable for MRI; however, they may degrade image quality in adjacent oral, sinus, or mandibular regions. Communicate with radiology to plan sequences that minimize artifact.
3. Coronary and Peripheral Vascular Stents
· Composition and magnetism: Most contemporary stents use stainless steel, nitinol (NiTi), or cobalt-chromium alloys—non- or weakly magnetic—with extremely low risk of displacement or deformation at 1.5 T and 3.0 T.
· Immediate MRI feasibility: Evidence indicates stents do not migrate at 3.0 T; MRI is generally feasible immediately after placement if clinically necessary.
· Heating: Measured temperature rise at 3.0 T from RF conduction is modest (about 2.6–4.7°C in test conditions). In vivo, convective blood flow substantially dissipates heat, making clinically relevant warming unlikely.
· Conservative practice: Many centers still recommend waiting up to 8 weeks post-implant to allow drug elution, endothelialization, and full tissue integration, balancing caution with clinical need.
Protocol guidance:
· Prefer 1.5 T when imaging near stented segments if artifact compromises diagnostic detail at 3.0 T.
· Leverage black-blood or turbo spin-echo sequences to reduce susceptibility effects.
4. Electronic Cardiac Devices: Pacemakers and ICDs
· Risk profile: In strong fields, pacing systems may experience lead-tip heating, inappropriate sensing, and output inhibition; severe cases risk myocardial burns or perforation and malignant arrhythmias.
· Traditional rule: Legacy non-MR-conditional pacemakers or implantable cardioverter-defibrillators (ICDs) were absolute contraindications to MRI.
· Modern nuance: Many contemporary systems are MR-Conditional with device-specific protocols (programming to MRI mode, SAR limits, field strength limits, monitoring). If a patient has any cardiac device, do not proceed without device identification and a standardized MR-Conditional workflow.
Bottom line:
· For non-MR-conditional pacemakers/ICDs, MRI is generally prohibited.
· For MR-Conditional devices, scan only under strict institutional protocol with cardiology and device rep support.
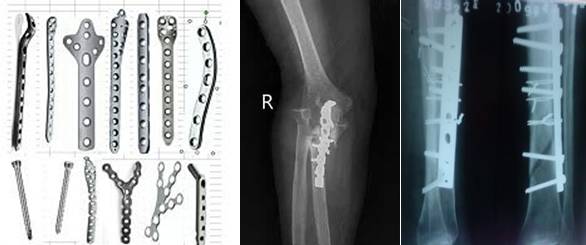
5. Orthopedic Internal Fixation: Plates, Screws, Rods, and Joint Implants
· Materials: Current internal fixation commonly uses pure titanium (e.g., Grade 2 CP Ti) or titanium alloys, with stainless steel and cobalt-chromium-molybdenum in some devices.
· Safety: Modern titanium systems exhibit negligible interaction with the magnetic field and do not migrate; MRI is typically permissible.
· Artifact: Titanium produces less susceptibility artifact than many stainless steel alloys, improving visualization of nearby soft tissues, bone marrow, and cartilage. Nevertheless, some distortion persists, especially at 3.0 T.
· Older hardware: Legacy ferromagnetic stainless steels can experience stronger forces and greater artifacts; these are often relative contraindications, particularly if hardware lies adjacent to major vessels or critical neural structures.
Imaging strategies for orthopedic metal:
· Prefer 1.5 T for regions with significant metal burden if diagnostic goals allow.
· Employ metal-artifact reduction techniques (high bandwidth, view-angle tilting, SEMAC/MAVRIC, thinner slices, STIR or Dixon fat suppression).
· Align frequency-encoding to displace artifact away from the region of interest when possible.
Patient and Provider Checklist
· Identify the implant: Make, model, material, and MR-Conditional status; carry the card to MRI.
· Match the scanner: If imaging near metal, consider 1.5 T; at 3.0 T, use artifact-reduction sequences.
· Control SAR: Follow device-specific limits; minimize RF duty cycle where required.
· Communicate: Surgeons, cardiologists, and radiologists should coordinate on risk, sequence selection, and monitoring.
· Post-scan: Assess for symptoms (heating, discomfort) and verify device function when applicable.
Special Considerations by Region
· Neurovascular clips/coils: Favor 1.5 T and spin-echo–based sequences for postoperative checks near the skull base.
· Maxillofacial: Remove removable dental appliances; tailor sequences to mitigate susceptibility around implants and orthodontic hardware.
· Cardiovascular: Early post-stent MRI is often feasible; use institutional policies for timing and SAR.
· Musculoskeletal: For titanium plates/screws, MRI is typically allowed; select MAR sequences to evaluate soft-tissue complications (infection, tendon pathology) around the implant.
· Pediatrics and oncology: Artifact minimization is key to lesion detection; MRI planning may combine ultrasound or CT when metal obscures targets.
Conclusion
Most contemporary implants made from Grade 2 CP titanium or titanium alloys are compatible with MRI when scanned under MR-Conditional guidelines. While the twin risks of displacement and excessive heating are minimal for titanium, imaging artifacts remain a practical concern—more so at 3.0 T than at 1.5 T. With careful device identification, thoughtful sequence selection, and inter-disciplinary communication, patients can safely undergo MRI while preserving diagnostic quality.
Frequently Asked Questions and Answers
Q1: Can titanium go in MRI?
A1: Yes. Titanium (including Grade 2 CP Ti) is non-ferromagnetic and typically labeled MR-Conditional or MR-Safe. When scanned within device-specific limits, it does not move or heat dangerously, though some image artifact may occur.
Q2: Is titanium safe in MRI?
A2: Generally yes. Titanium’s weak magnetic susceptibility and low RF heating profile make it safe under standard protocols at 1.5 T and 3.0 T. Always confirm the implant’s MR-Conditional labeling and follow SAR and sequence guidelines.
Q3: Can you have an MRI with a titanium knee replacement?
A3: In most cases, yes. Titanium knee components are MRI-compatible, but they can cause artifacts near the joint. Radiology teams often prefer 1.5 T and employ metal-artifact reduction sequences to evaluate surrounding soft tissues and bone effectively.


