Nickel titanium shape memory alloys (SMAs), commonly referred to as nickel titanium or titanium nickel alloys, have revolutionized the field of orthopedic implants due to their unique mechanical and functional properties. Composed predominantly of nickel and titanium, these alloys exhibit a remarkable phenomenon known as the shape memory effect, allowing the material to return to a pre-defined shape upon heating. This property, combined with superelasticity, excellent mechanical strength, corrosion resistance, and good biocompatibility, positions nickel titanium alloys as a preferred material for a wide range of orthopedic applications.
At the heart of these unique behaviors are two distinct crystal structures—austenite and martensite—which the alloy adopts at different temperatures. Austenite, stable above the transformation temperature, is characterized by high stiffness and resistance to deformation, whereas martensite, stable below the transformation temperature, is relatively soft and can be easily shaped. These phase-dependent properties enable the design of implants that can adapt to complex physiological environments and loading conditions.
This article systematically reviews the application of nickel titanium shape memory alloys in orthopedic implants, explores the influence of phase transformation temperatures and key mechanical indicators, and discusses the development trends and future directions of these advanced biomaterials
1. Applications of Nickel Titanium Implants in Orthopedics
Nickel titanium alloys have found widespread clinical acceptance in orthopedic surgery, particularly for their capacity to provide reliable, adaptive fixation and promote rapid functional recovery. The following subsections illustrate the main types of titanium nickel orthopedic implants and their clinical significance.
1.1 Application of Nickel Titanium Embracing Devices
The nickel titanium memory alloy embracing device is commonly utilized in the treatment of multiple rib fractures—a condition that accounts for 10% to 26% of all thoracic injuries and presents significant therapeutic challenges. Multiple rib fractures can result in serious chest trauma, including flail chest, characterized by paradoxical movement and compromised respiratory function.
Clinical studies involving 120 patients with traumatic flail chest provide compelling evidence for the superiority of nickel titanium embracing devices over traditional conservative management. In these studies, 60 patients received conservative treatment, while the other 60 underwent internal fixation with shape memory alloy embracing devices. Outcomes demonstrated that the embracing device group enjoyed markedly shorter recovery times across various indicators, including normalization of abnormal heart rhythms and inflammatory markers. Importantly, these patients also experienced a more stable and rapid restoration of physical function.
The excellent tissue compatibility of nickel titanium embracing devices is a key factor in their clinical utility, minimizing the risk of adverse reactions and supporting the healing of traumatized tissues. Moreover, these devices have proven highly effective in the treatment of metacarpal and metatarsal fractures. In a series of 65 patients treated for metacarpal or metatarsal shaft fractures with nickel titanium embracing devices, all fractures healed without incidences of nonunion, malunion, delayed union, or infection, underscoring both the reliability and safety of these implants.
1.2 Application of Nickel Titanium Patella Concentrators
Patellar fractures are relatively common, often resulting from direct trauma or violent impact. Failure to provide timely and effective treatment can result in permanent damage to the knee joint and significant, potentially lifelong disability. The nickel titanium shape memory patella concentrator has shown outstanding efficacy in the fixation of comminuted patellar fractures.
A study involving 63 patients with transverse patellar fractures compared surgical outcomes between two groups: 29 patients underwent open reduction and internal fixation using the memory alloy patella concentrator, while 34 patients received conventional tension band wiring. The group treated with the patella concentrator demonstrated shorter operative times, higher intraoperative safety, and, most importantly, faster and more complete functional recovery of the knee joint. The superelasticity and adaptability of the titanium nickel alloy ensure stable fixation, minimize soft tissue irritation, and facilitate early postoperative mobilization.
1.3 Application of Nickel Titanium Memory Alloy Spinal Rods
The advent of nickel titanium spinal instrumentation has been a watershed in the management of spinal disorders, particularly in cervical spondylosis and other deformities requiring precise correction. Memory alloy rods, pre-bent to a therapeutic configuration, can be implanted and subsequently activated to achieve targeted spinal realignment.
Compared with traditional materials, nickel titanium memory alloys offer superior mechanical properties—including enhanced tensile, torsional, and impact resistance—as well as exceptional wear resistance. These features translate into better long-term durability, reduced risk of implant failure, and a lower incidence of debris-induced tissue damage. In orthopedic cases where complex, stable fixation is required—such as in severe deformities or in osteoporotic patients—nickel titanium memory alloy rods provide robust, effective internal support.
1.4 Other Applications in Orthopedic Implants
Beyond embracing devices, patella concentrators, and spinal rods, titanium nickel shape memory alloys have been employed in a diverse array of orthopedic implants, such as:
· Bow-tooth nails
· Skull bone claws
· Scaphoid internal fixation forks
· Wrist fusion devices
These implants are primarily used in the treatment of fractures of the ribs, metatarsals, patella, and in various spinal deformities. Extensive clinical research consistently demonstrates that nickel titanium shape memory alloy implants outperform conventional fixation devices, offering improved healing rates, greater biomechanical stability, and fewer complications.
2. Phase Transformation Temperature of Nickel Titanium Shape Memory Alloy Implants
The extraordinary functional properties of nickel titanium SMAs—especially shape memory and superelasticity—are fundamentally governed by their phase transformation temperatures. Precise control of these thermal properties is essential for ensuring reliable clinical performance.
2.1 Importance of Accurate Measurement
Due to limitations in the accuracy of compositional analysis for nickel and titanium, conventional measurement methods are insufficient to guarantee the desired shape memory or superelastic properties. Therefore, thermal analysis techniques (such as differential scanning calorimetry) and equivalent bending-free recovery methods are employed to accurately determine the transformation temperatures of SMAs.
2.2 Effect of Annealing Temperature
Among the many factors influencing phase transformation temperature, annealing temperature exerts a significant effect, generally exhibiting a positive correlation within a certain range. Higher annealing temperatures tend to raise the transformation temperature, as changes in precipitate phases and grain size during rapid thermal annealing alter the material’s response to temperature. This relationship must be carefully managed to achieve the intended intraoperative and in vivo performance of the implant.
2.3 Effect of Alloy Composition
Alloy composition—specifically, the precise ratio of nickel to titanium—is another critical determinant of phase transformation temperature. Even minor variations in composition can shift the transformation temperatures, potentially impacting the shape memory or superelastic behavior of the implant. Thus, rigorous compositional control and post-processing are indispensable in the manufacturing of high-performance titanium nickel orthopedic devices.
3. Selection of Shape Memory Effect or Superelasticity Based on Clinical Requirements
The clinical environment dictates whether the shape memory effect or superelasticity of nickel titanium alloys is most desirable. The transformation temperature at which the material exhibits these properties, particularly the Af point (austenite finish temperature), is carefully engineered according to the intended application.
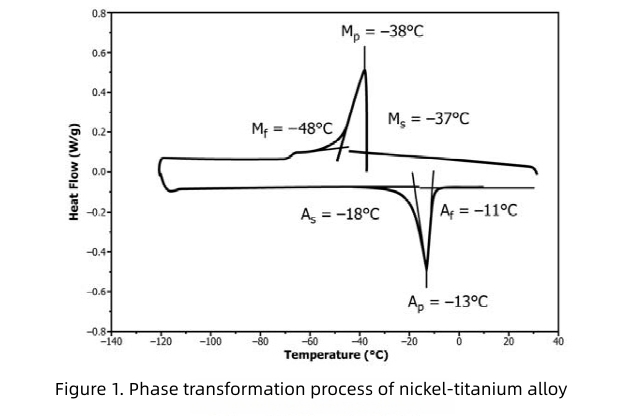
· For superelasticity at room temperature, the Af point should be controlled below 22–25°C.
· For superelasticity within the human body, the Af point must be below 37°C.
Most orthopedic implants are designed with a transformation temperature below 37°C, ensuring that, once implanted, the device undergoes a martensite-to-austenite phase transformation upon exposure to body heat. This transition imparts superelasticity, enabling the implant to adapt to physiological movement and loading.
The ending temperature of the martensite-to-austenite reverse transformation is influenced by both annealing temperature and duration. Higher solution temperatures, combined with longer annealing times, can first lower and then raise the Mp temperature. Cooling rate also plays a significant role; as the cooling rate decreases, the Ms and Mf transformation temperatures decline, while slower cooling can elevate the M–A austenite transformation range. These thermal characteristics must be meticulously tailored to ensure optimal implant function and long-term reliability.
4. Impact of Key Indicators on Shape Memory Alloy Implant Performance
Given their increasing deployment as Class II and Class III medical devices, most nickel titanium orthopedic implants are intended for long-term or even permanent implantation. As such, several critical indicators must be rigorously controlled to ensure both safety and efficacy.
4.1 Biocompatibility
Nickel titanium alloys exhibit excellent biocompatibility, which is crucial for minimizing inflammatory responses and ensuring harmonious integration with bone and soft tissue. The elastic modulus of these alloys is close to that of human bone, reducing the risk of stress shielding and promoting natural bone remodeling.
4.2 Superelasticity and Mechanical Properties
Superelasticity is perhaps the most valued property of titanium nickel shape memory alloys, enabling implants to accommodate physiological movements without permanent deformation. These alloys also exhibit low magnetic susceptibility, high wear resistance, corrosion resistance, and outstanding fatigue life. Such properties are especially important in load-bearing implants, where mechanical integrity over thousands or millions of cycles is required.
4.3 Key Performance Indicators
· Chemical composition: Directly influences phase transformation temperature, mechanical strength, and corrosion resistance.
· Transformation temperature: Determines whether the implant will function as intended under physiological conditions.
· Mechanical performance: Critical for effective fixation; failure to meet specifications can result in complications such as implant loosening, fracture, or poor bone healing.
Rigorous adherence to these indicators is essential. Any deviation can compromise the implant’s performance, potentially leading to adverse clinical outcomes such as nonunion, malunion, or implant failure.
5. Developmental Trends and Future Directions of Nickel Titanium Shape Memory Alloy Implants
The ongoing evolution of nickel titanium shape memory alloy technology is driven by both materials science and clinical innovation. Several key trends are shaping the future of these advanced biomaterials:
5.1 Alloy System Diversity and Optimization
To realize the shape memory effect, at least ten basic alloy systems have been developed—if all alloying elements are considered, the number of possible compositions rises into the hundreds. The most widely used in clinical practice are titanium-based alloys, copper-based alloys, and iron-based alloys. Among these, titanium nickel shape memory alloys are the most thoroughly researched and have the most advantageous memory properties.
5.2 Addressing Biocompatibility and Nickel Ion Release
While the mechanical and functional properties of titanium nickel alloys are outstanding, the presence of nickel—a potential allergen and toxin—raises concerns about long-term biocompatibility. Nickel ion release from the implant surface can induce cytotoxicity, allergic reactions, or other adverse effects. To counteract this, contemporary research focuses on surface modification techniques such as:
· Biocompatible coatings (e.g., titanium oxide, hydroxyapatite)
· Ion implantation
· Surface passivation
These methods effectively inhibit nickel ion release, significantly improving the safety profile of titanium nickel implants.
5.3 Matching Elastic Modulus to Bone
A major advantage of nickel titanium shape memory alloys is their elastic modulus, which closely matches that of cortical bone. This compatibility reduces the risk of stress shielding—a phenomenon whereby a stiffer implant absorbs excessive load, leading to bone resorption and weakening.
5.4 Integration with Advanced Technologies
The convergence of shape memory alloy technology with virtual reality (VR) and artificial intelligence (AI) is opening new frontiers in orthopedic implant design and surgical planning. VR enables precise preoperative modeling and simulation, while AI can be used to optimize implant geometry and predict postoperative outcomes. These tools promise to further enhance the safety, efficacy, and personalization of titanium nickel orthopedic solutions.
5.5 Regulatory and Clinical Perspectives
Given their status as high-risk, long-term implants, titanium nickel devices are subject to strict regulatory oversight. Ongoing post-market surveillance, clinical trials, and material certification processes are essential to ensure their continued safety and efficacy.
Conclusion
Nickel titanium shape memory alloys have emerged as a cornerstone of modern orthopedic implant technology, offering unparalleled functional properties, biocompatibility, and adaptability. Their unique phase transformation behavior enables both shape memory and superelasticity, facilitating innovative solutions for complex orthopedic challenges—from rib and patella fractures to spinal deformities and beyond.
The continued refinement of alloy composition, phase transformation temperature, and surface modification techniques is enhancing the clinical performance and safety of these implants. As research progresses, and as new technologies such as VR and AI are integrated into implant design and surgical practice, the future of nickel titanium shape memory alloy orthopedic implants appears both promising and transformative.
Frequently Asked Questions and Answers
1. What are the primary advantages of nickel-titanium shape memory alloys over conventional materials (like stainless steel or titanium alloys) for orthopedic implants?
Nickel-titanium (titanium nickel) shape memory alloys offer several key advantages over traditional materials such as stainless steel or conventional titanium alloys. Their shape memory effect allows for minimally invasive implantation and adaptive fixation, as the implant can be inserted in a compact form and then expanded or conformed in situ. Superelasticity enables the implant to accommodate dynamic physiological movements and loads without permanent deformation, reducing the risk of implant failure. Additionally, the elastic modulus of nickel titanium alloys closely matches that of bone, minimizing stress shielding and promoting healthy bone remodeling. These alloys also exhibit excellent corrosion resistance, wear resistance, and long-term fatigue strength, making them particularly suitable for demanding orthopedic applications.
2. Which types of orthopedic implants most commonly use nickel-titanium shape memory alloys, and why are these alloys uniquely suited for those specific applications?
The most common orthopedic implants made from nickel-titanium shape memory alloys include embracing devices for rib fractures, patella concentrators for knee injuries, and spinal rods for deformity correction. These alloys are uniquely suited to such applications due to their ability to provide stable, adaptive fixation while accommodating anatomical movements and changes in loading. Their superelasticity and shape memory effect facilitate secure bone alignment and rapid, functional recovery, even in complex or high-mobility anatomical sites. Other applications include bone claws, internal fixation forks, and fusion devices for small bones and joints.
3. Are nickel-titanium shape memory alloys safe for long-term orthopedic implant use, and how are potential risks (such as nickel leaching or allergic reactions) mitigated in clinical practice?
Nickel-titanium shape memory alloys have demonstrated excellent long-term safety and biocompatibility in clinical use. However, the risk of nickel ion release and potential allergic reactions remains a concern. To mitigate these risks, manufacturers employ advanced surface modification techniques such as applying biocompatible coatings, passivation, or ion implantation to create a stable barrier that prevents nickel leaching. Extensive preclinical and clinical testing, along with rigorous regulatory standards, ensure that these implants meet stringent safety requirements for long-term implantation. Ongoing research continues to refine these approaches to further enhance the biocompatibility of nickel titanium orthopedic implants.


