Anodized titanium is a commonly used method for surface modification of titanium and titanium alloys. It involves using a titanium workpiece as the anode and aluminum or stainless steel as the cathode, forming a thin oxide film with good insulation, wear resistance, and corrosion resistance on the surface of the titanium workpiece by electrolysis. This oxide film not only protects the workpiece but also provides a distinct decorative effect, making anodized titanium highly valued in both industrial and consumer applications. Compared to other surface coloring technologies such as electroplating, electroless plating, PVD coating, electrolytic coloring, spraying, high-temperature oxidation, and micro-arc oxidation, anodizing stands out due to its simple operation, low cost, and the ability to produce dense and uniform films. These advantages have led to the widespread industrial adoption of anodized titanium for products requiring both functional performance and aesthetic appeal. However, the final color of anodized titanium is influenced by various factors, including voltage, electrolyte composition, temperature, and processing time. Understanding these factors is critical for achieving consistent and desirable coloration. This article reviews current research on the key factors affecting the color change of anodized titanium, with the aim of optimizing surface treatment processes for enhanced performance and design flexibility.
1. Principle of Color Formation in Anodized Titanium
The coloration of anodized titanium arises from the optical properties of the oxide film formed during anodization. When a titanium or titanium alloy is subjected to an external electric field during the anodizing process, a dense oxide layer—ranging from a few nanometers to several hundred nanometers in thickness—forms on the metal surface. This thin oxide film is generally transparent and exhibits strong reflective and refractive effects on incident light. The phenomenon behind the color formation is known as thin-film interference.
When light strikes the anodized surface, some of it is reflected from the outer surface of the oxide film, while the rest penetrates the film, reflects off the interface between the oxide layer and the underlying metal, and then exits the film. Depending on the thickness of the oxide layer, the light waves combine constructively or destructively, amplifying certain wavelengths and diminishing others. This interference creates the characteristic colors seen on anodized titanium surfaces.
The specific color observed is determined primarily by the thickness of the oxide film, which controls which wavelengths are reinforced or suppressed. As the thickness increases, the color shifts in sequence: yellow → purple → blue → light blue → silver → yellow → pink → purple → cobalt blue → green → yellow/green → pink → green, and so on. This sequence is widely documented in both laboratory and industrial environments, and it forms the basis for the controlled colorization of titanium products through anodizing.
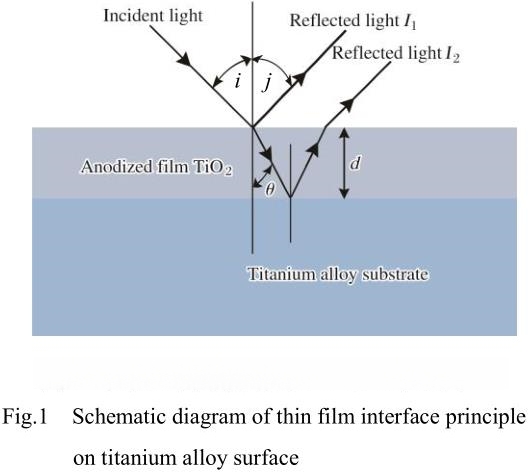
formula for calculating optical path difference
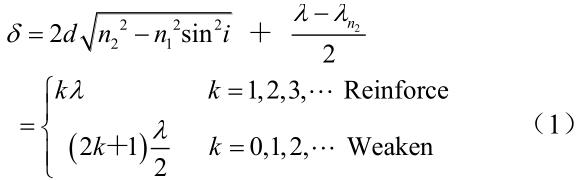
In the formula: λ represents the optical path difference between the reflected light from the upper and lower surfaces of the film; d represents the thickness of the oxide film; n1 and n2 are the refractive indices of air and the oxide film, respectively (n1~1, n2 is 2.2~2.7); i is the angle of incidence; and λ is the wavelength of the incident light. When the optical path difference is equal to or close to an even multiple of λ/2 wavelength, both light beams are strengthened; when the optical path difference is equal to or close to an odd multiple of λ/2 wavelength, both light beams are weakened. Due to the high refractive index of the titanium oxide film, the half-wave loss of the beam reflected from the lower surface of the film cannot be ignored.
2. Factors Affecting the Color of Anodized Titanium Films
The ultimate color of an anodized titanium surface is determined by several key factors, both controllable and inherent to the material. Understanding and controlling these factors is essential not only for achieving specific aesthetic results but also for meeting the performance and quality requirements outlined in relevant Titanium Standard documents.
2.1 Anodizing Voltage
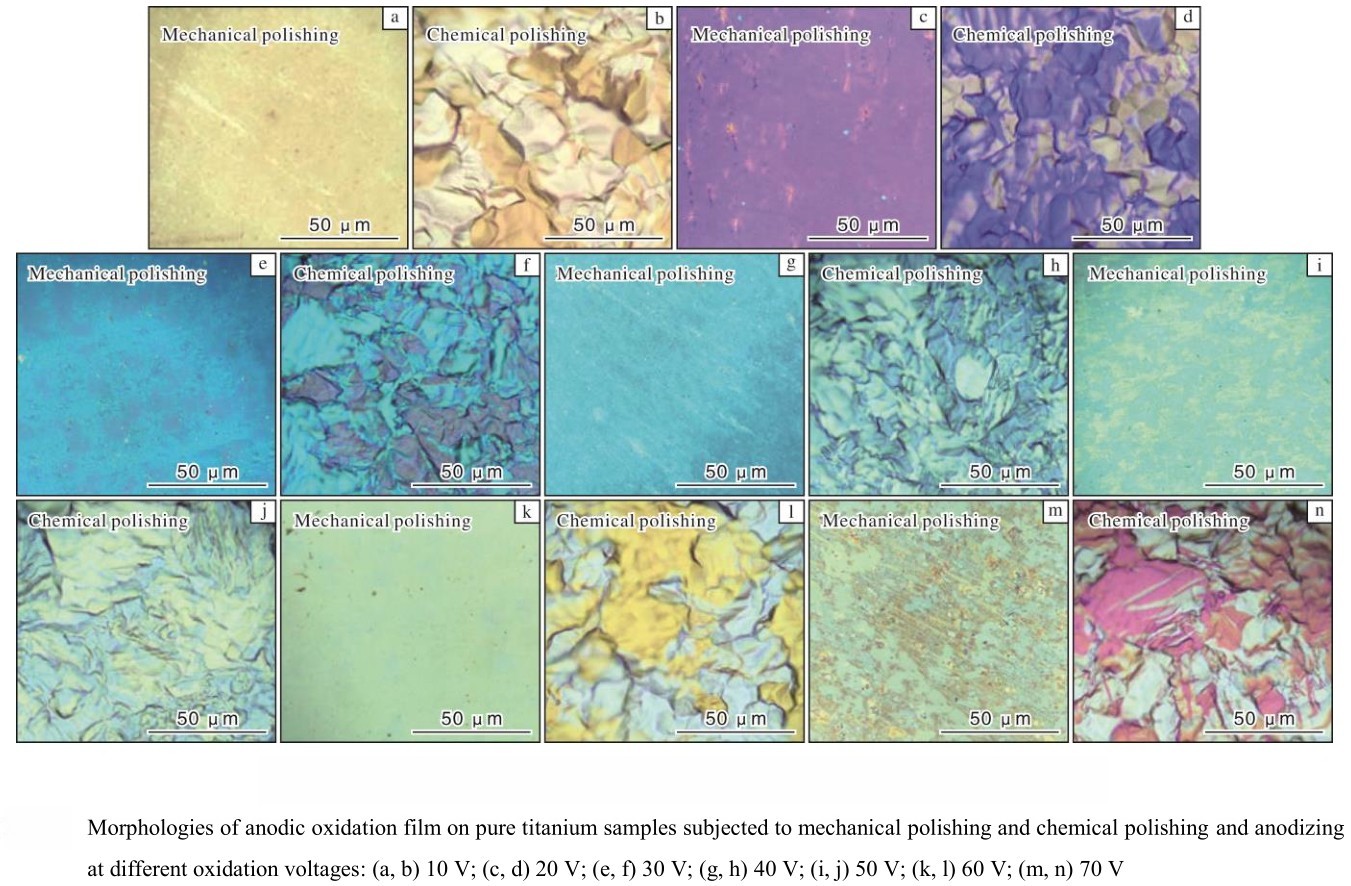
Among all the variables, the anodizing voltage is the most critical factor influencing the color of the titanium oxide film. During the anodizing process, oxygen ions are generated and migrate toward the titanium surface, where they react with titanium cations to form a dense oxide film. The electric field strength, determined by the applied voltage, directly affects the migration rate of oxygen ions and thus the growth rate and ultimate thickness of the oxide layer.
There is a linear relationship between the anodizing voltage and the oxide film thickness; as the voltage increases, the film becomes thicker. Research has shown that for every 1V increase in voltage, the film thickness increases by approximately 2.0 nm. As the thickness changes, so do the refractive index, reflectivity, and light flux of the film, resulting in different interference colors. By precisely controlling the anodizing voltage, manufacturers can reliably produce a wide range of colors on titanium surfaces. This method is widely used for both decorative and functional purposes in medical, consumer, and aerospace products.

2.2 Anodizing Time
Anodizing time is another key parameter that influences the color of the oxide film. In the early stages of anodizing, the oxide layer grows rapidly. The longer the anodization time, the more oxide accumulates on the titanium substrate, and the thicker the film becomes. However, as the process continues, the film growth rate slows, and the crystal grains within the film grow larger and denser.
Interestingly, while longer anodizing times can initially increase color saturation, excessive duration leads to a decrease in color vibrancy. This reduction in saturation is due to the increased density and size of the crystalline grains, which obscure the underlying film and alter the light interference effects. Furthermore, if the anodizing time is too short, the oxide film may not be stable or uniform, making it more susceptible to contamination and resulting in inconsistent coloration. Therefore, optimizing anodizing time is essential for achieving both the desired color and film stability.
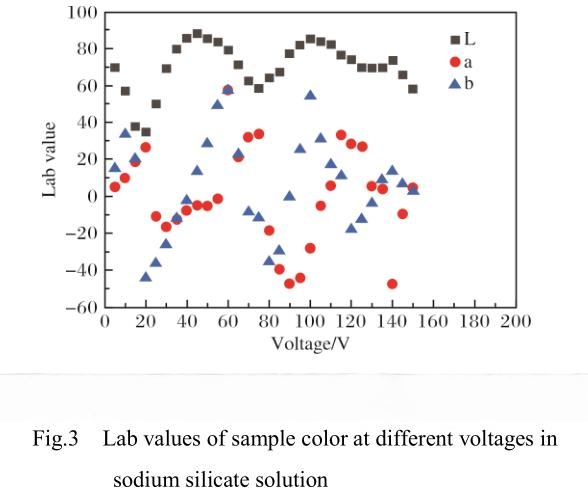
2.3 Electrolyte Composition
The choice of electrolyte plays a substantial role in the anodizing process and the resulting color of the oxide film. Most studies and industrial applications use acidic or near-neutral salt solutions, as alkaline solutions tend to dissolve the oxide layer too aggressively, making process control difficult. For instance, medical-grade pure titanium anodized in 5.0% citric acid solution shows improved corrosion resistance and a broad spectrum of surface colors.
Different electrolytes can produce different film formation rates and colors under the same voltage and anodizing time. For example, when anodizing Ti-6Al-4V alloy at 20V, using oxalic acid as the electrolyte yields a purplish-red film, while using phosphoric acid under identical conditions results in a purple film. Such differences are attributed to the varying dissolution rates and ionic interactions of the electrolytes with the titanium surface. The electrolyte not only affects the growth rate and thickness of the oxide film but also its structure and uniformity, thereby influencing the final color. Selecting the appropriate electrolyte is crucial for both color consistency and functional properties of anodized titanium.
2.4 Anodizing Temperature
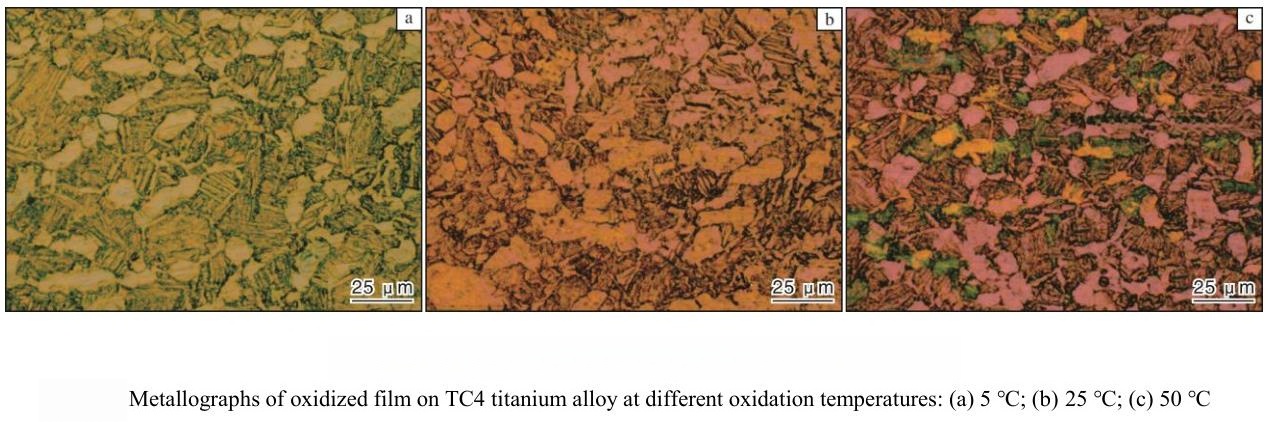
Temperature is a fundamental parameter in all electrochemical oxidation reactions, including anodizing. In the case of titanium alloys, higher temperatures can accelerate the migration of ions and the overall anodizing reaction, potentially increasing the oxide film growth rate. However, as temperature rises—especially above 40°C—the average color deviation (ΔE) of the oxide film increases, leading to less uniform coloration.
This phenomenon occurs because elevated temperatures not only facilitate ion migration but also increase the rate at which the electrolyte attacks the growing oxide film. When the local rate of corrosion exceeds the rate of oxide formation, the result is an uneven and patchy film. Thus, while moderate temperature increases can be beneficial, excessive temperatures are detrimental to color uniformity and film quality. Careful temperature control is therefore necessary to balance process efficiency and product consistency in anodized titanium applications.
2.5 Other Influential Factors
In addition to the primary parameters listed above, several secondary factors can influence the coloration of anodized titanium. These include the texture orientation and pre-treatment of the titanium substrate. For example, anodizing Ti-6Al-4V alloy samples in 1M sulfuric acid demonstrates that different substrate grain orientations result in different oxide film thicknesses and, consequently, different colors.
X-ray reflectivity studies have shown that oxide films grown on basal (0001) planes can reach thicknesses of around 120 nm (yellow color), while those on transverse (2110) planes may reach about 140 nm (pink color). Since the orientation of the grains affects the local growth rate of the oxide film, the resulting color distribution may be non-uniform across the titanium surface. Pre-treatment methods such as polishing, etching, or surface cleaning also impact the initial surface state, which in turn affects the adhesion, growth, and appearance of the oxide film. To achieve high-quality, uniform coloration, it is essential to consider these additional factors in both laboratory research and industrial practice.
3. Conclusion
Anodizing is a vital surface treatment process for imparting color to titanium and titanium alloys, combining aesthetic appeal with enhanced wear, corrosion, and insulation properties. Products featuring anodized titanium are now widely used in aerospace, petrochemical, biomedical, and consumer decorative applications. Research and industrial experience demonstrate that the primary factors affecting the color of anodized titanium oxide films are anodizing voltage, anodizing time, and electrolyte temperature. Secondary influences include electrolyte composition, substrate material, texture orientation, and pretreatment methods.
A comprehensive understanding of these factors—supported by robust scientific principles and adherence to relevant Titanium Standard specifications—enables precise control over the color and performance of anodized titanium products. This not only meets the needs of modern industry but also opens new possibilities for design and innovation in both functional and decorative applications.
Frequently Asked Questions and Answers
1. What are the primary factors that influence the color variation of titanium during anodizing processes?
The primary factors include anodizing voltage, anodizing time, electrolyte composition, and temperature. Secondary influences such as substrate texture orientation and pre-treatment also affect the final color. Each factor impacts the thickness and optical properties of the oxide film, which together determine the observed color.
2. How does the thickness of the oxide film affect the final color of anodized titanium surfaces?
The color is produced by thin-film interference, which depends on the oxide film’s thickness. As the film thickens, it selectively reinforces or cancels out certain wavelengths of light, resulting in a predictable sequence of colors. Precise control of voltage and processing time allows for consistent and repeatable color results.
3. What role do electrolyte type and operating temperature play in determining the color of anodized titanium?
Different electrolytes affect the growth rate, structure, and uniformity of the oxide film, leading to variations in color under similar processing conditions. Temperature influences ion migration and the rate of the anodizing reaction; higher temperatures can accelerate oxide formation but may also increase non-uniformity and patchiness if not properly controlled.


